For the reliable and safe production and delivery of high-quality radiopharmaceutical products our production sites maintain production environments in conformity with the latest Good Manufacturing Practice (GMP). In addition to production sites in Chiba (Sodegaura) and Hyogo (Sanda) for SPECT products we established PET laboratories across the country for our entry into the PET business. All of our employees demonstrate professionalism and are devoted day and night to ensuring excellent quality keeping in mind the maintenance and enhancement of a clean environment for pharmaceutical production as well as strict environmental and safety controls adequate for RI-handling facilities. Nihon Medi-Physics designates one of the corporate functions specifically to be engaged in supervising product quality and safety according to relevant regulations such as the Pharmaceutical Affairs Law GQP (Ministerial Ordinance on standards for Quality Assurance for Drugs Quasi-drugs Cosmetics and Medical Devices) and GVP (Ministerial Ordinance Related to Standards for Post-Marketing Safety Management of Drugs Quasi-drugs Cosmetics and Medical Devices). For an enterprise involved in the special field of nuclear medicine proper and strict control of radiation is the foremost priority. Nihon Medi-Physics has established a department responsible for RI management where dedicated staff members including radiation protection supervisors having special knowledge and experience are assigned. To secure the safety of workers and surrounding areas against radiation the RI management department controls exposure to workers and provides safety controls for work rooms and other environments. As an example of these measures radioactivity inside and outside the facility is measured once a month to monitor the condition of radioisotope contamination in the production site of radiopharmaceuticals. Nihon Medi-Physics establishes and applies in-house standards that are stricter than the management criteria of regulations. We periodically implement necessary safety measures and examine work processes and environmental conditions to conduct safety radiation management on a daily basis.
 Chiba Facility
Chiba Facility Hyogo Facility
Hyogo Facility PET Laboratory
PET LaboratorySafety management system for product quality
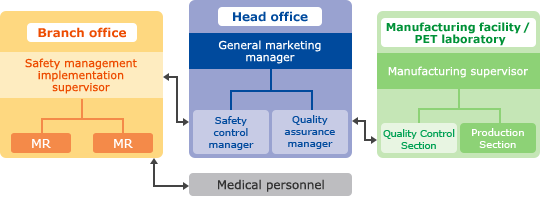 Quality assurance & Safety management system
Quality assurance & Safety management systemRadiation safety management system
![]()
About the contents of this website
Product information in this Website is provided to offer general information on the Company’s pharmaceutical products. It is not intended to provide medical advice or usage descriptions of the Company’s pharmaceutical products. This Website is not to be used as an alternative for medical practitioners such as physicians and pharmacists to consult with patients in order to determine medical treatments. In addition, the information about the products (including products under development) on this Website does not constitute a promotion or commercial advertisement of the product or its efficacy.
Product information provided on this Website is intended for medical practitioners for use in Japan, and the contents therein are based on approvals that have been granted in Japan. If you are accessing this Website from outside of Japan, please be aware that the product information available on this Website might not be appropriate for you. Please confirm the approval status and content in your relevant country.